Two research teams from the University of Bern and the ETH Zurich have developed a new method to shed light onto a mostly unknown process of bacterial protein production. Their results could be used for the design of new antibiotics.
A possible way to new antibiotics
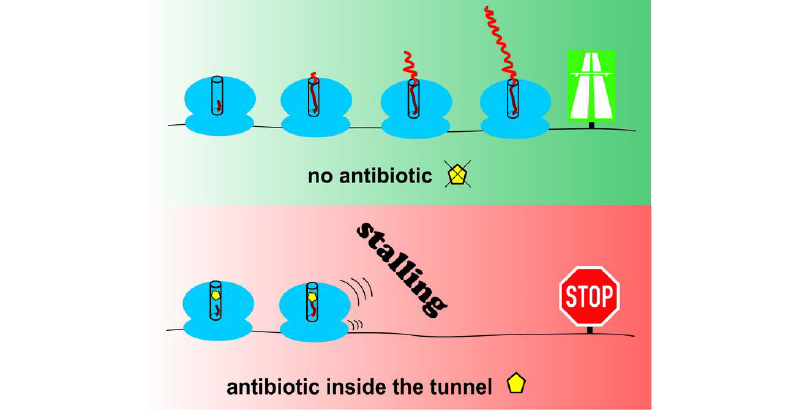
Ribosomes are the factories of the cell and, as such, are responsible for the fabrication of proteins. They resemble a complex and highly dynamic machinery made of ribosomal RNA molecules and a variety of ribosomal proteins. As long as theses factories run smoothly, amino acids are put together incessantly deep in the center of the complex to form new proteins that are then released through a special narrow ribosomal exit tunnel. But sometimes things get stuck in the fabrication line. Proteins are hindered to leave the exit tunnel and the whole machinery is stalled.
Widespread stalling is fatal for the cell, and that is why it is efficiently targeted by antibiotics. Indeed, ribosomes are so central for the functioning of the cell that any serious obstruction of the machinery has serious consequences for the whole organism. That is why more than half of the naturally occurring antibiotics target the ribosome in some way. Understanding the mechanisms of where and how these antibiotics obstruct the protein factory is therefore a promising path towards new antibiotics, a research field that is becoming more and more important as the known antibiotics gradually lose their potency. "We need a targeted use of new antibiotics in order to fight antibiotics resistances", says Norbert Polacek of the Department of Chemistry and Biochemistry at the University of Bern and group leader of in the National Centre of Competence in Research
"RNA & Disease". Together with the group of Jonathan Hall of the Department of Chemistry and Applied Biosciences at the ETH Zurich, he has now discovered a possible way to new antibiotics. The findings of the study have been published in "Nucleic Acids Research".
"Brake application" described
Ribosome stalling is a promising starting point for this kind of research, as the functional role of the ribosomal exit tunnel to polypeptide synthesis and protein folding is only beginning to be understood in molecular terms. It seems like the tunnel acts as something of a brake pedal of the translation process, building up proteins from according messenger RNA sequences. Sometimes the hindering is just slowing down the translation, sometimes the specific interactions of the nascent proteins with the tunnel wall result in a complete translation arrest, the so-called ribosomal stalling. But how exactly does this arrest happen? And how do the stations further down the assembly line know that work needs to rest?
In order to answer this, the research groups examined the stalling process induced by erythromycin and other macrolide antibiotics (which inhibit the protein synthesis of bacteria). They figured out an ingenious way to alter tiny parts of the tunnel and to see how this affected stalling By replacing single nucleobase functional groups or even single atoms of ribosomal RNA they were able to demonstrate the importance of specific functional groups in the tunnel for drug-dependent ribosome stalling. They identified the exact parts of the ribosomal machinery responsible for sensing and transmitting the stalling signal from the exit tunnel back to the peptidyl transferase center of the ribosome where the amino acids are linked together to build up proteins. These nucleobases do not contribute significantly to the overall mechanism of protein biosynthesis, yet their elaborate role for co-translational monitoring of nascent peptide chains inside the exit tunnel can explain their evolutionary conservation.
Joining forces for complex problems
The research is not only interesting for the pharmaceutical implications, but also because it highlights the potential of interdisciplinary approaches to complex molecular biology problems. "The work could not have been accomplished by either group in Bern or Zürich alone", Norbert Polacek stresses. Both groups had to contribute their expertise, the Hall group from the ETH Zurich the competence in RNA chemical synthesis, and the Polacek group from the University of Bern the skills in ribosome biochemistry. In this, the research is a perfect example of the research philosophy of the NCCR "RNA & Disease – The Role of RNA Biology in Disease Mechanisms", bridging gaps between disciplines to deepen our understanding of the various connections between the RNA and disease.
Publication details:
M. Koch, J. Willi, U. Pradere, J. Hall and N. Polacek (2017) Critical 23S rRNA interactions for macrolide-dependent ribosome stalling on the ErmCL nascent peptide chain Nucleic Acids Res, 45(11), 6717-6728
https://academic.oup.com/nar/article/45/11/6717/3076973